Plunkett Research Group
Southern Illinois University
Our group utilizes self-assembly motifs and the tools of synthetic and physical organic chemistry to address problems at the interface of chemistry and materials science. Our target materials are 1) novel semiconductors for organic photovoltaic devices, 2) highly ordered conjugated polymers, and 3) smart surfaces for biomacromolecule immobilization.
Research Topics:
1. CP-PAH Based Electron Acceptors
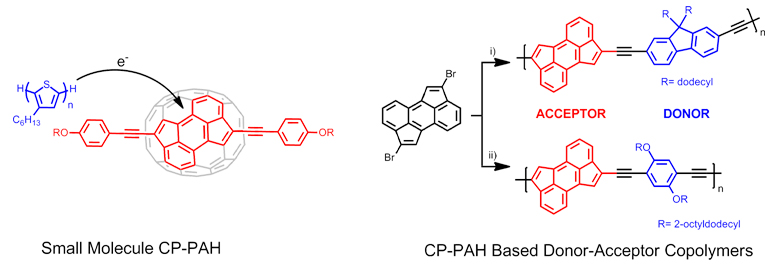
Although cyclopenta-fused polycyclic aromatic hydrocarbons (CP-PAHs) have been synthesized for many decades to provide compounds useful to elucidate PAH aromaticity or to create geodesic structures, very few CP-PAHs have been utilized as active components in electronic devices. Our group is interested in developing new synthetic methods to access the scalable synthesis of novel CP-PAHs that possess unique photochemical and electrochemical properties. The defining feature of these materials is their ability to accept electrons owing to the formation of cyclopentadienyl anion-like substructures upon reduction. Our recent work (J. Am. Chem. Soc. 2012, 134, 15783-15789 and Org. Lett. 2013, 15, 1202-1205) utilizes a palladium-catalyzed cyclopentannulation transformations to access small molecule electron acceptors based on functionalizable cyclopenta[hi]aceanthrylenes and dicyclopenta[de,mn]tetracenes. The synthetic pathways are straight-forward and allows functionalization of the CP-PAHs on either the 5-membered rings or the 6-membered rings to access differing electrochemical properties. We have further utilized these CP-PAH based electron acceptors in donor-acceptor copolymers (Angew. Chem. Int. Ed., 2012, 51, 12321-12324) and have accessed low band gap (~1.3 eV) materials. We continue to explore these materials (Syn. Lett., SYNPACTS article, 2013, 24, 898-902), as well as other CP-PAH systems, as active components in organic electronic devices. This work is now funded by a NSF CAREER grant (NSF CHE-1352431).
2. Highly Ordered Polymer Nanostructures
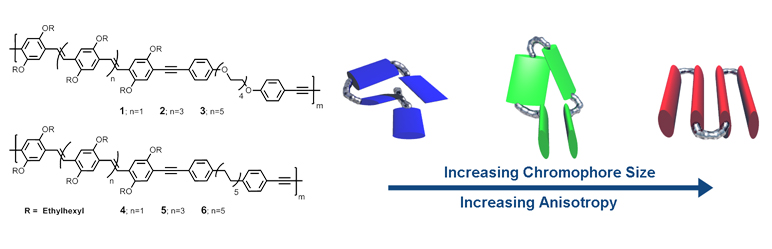
In collaboration with David Vanden Bout's group at the University of Texas at Austin, we are currently investigating the role of backbone and side-chain composition in conjugated polymers (CP) on the morphology at a single polymer level. We have developed methods to create well-defined polychromophore polymers (Macromolecules, 2012, 45, 5051-5057) composed of rigid phenylene-vinylene (PPV) chromophores and flexible segments. Through stepwise synthetic transformations, we employ strict control over the chromophore length and have found the subsequent polymers possess drastically different anisotropies via Single Molecule Fluorescence Polarization Spectroscopy (J. Phys. Chem. Lett., 2013, 4, 2520-2524). While trimer based PPVs are isotropic and quite disordered, septamer based PPVs are highly anisotropic and possess chromophores that are more aligned than even native MEH-PPV polymers. We are expanding on these initial investigations to look at the role of side-chain regioregularity (J. Org. Chem., 2014, 79, 7093-7102), H-bonding motifs, and methods that can translate high degrees of order from the single polymer regime to bulk films.
3. Smart Surfaces
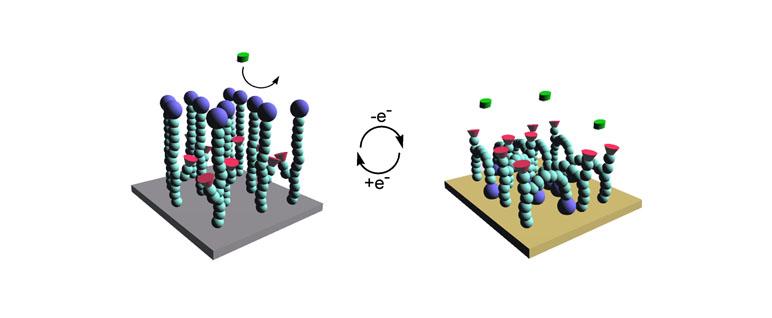
Surfaces that can reversibly immobilize biomolecules or cells are of great importance to biotechnology applications. To date, most immobilization techniques simply use the switching of a surface from hydrophilic to hydrophobic. Although these systems have shown promise, they lack the ability to specifically immobilize macromolecules. We are currently working on new reversible ligating surfaces that can immobilize specific biomacromolecule by varying the surface electrostatic potential.